A close look at diabetic retinopathy: preventing blindness
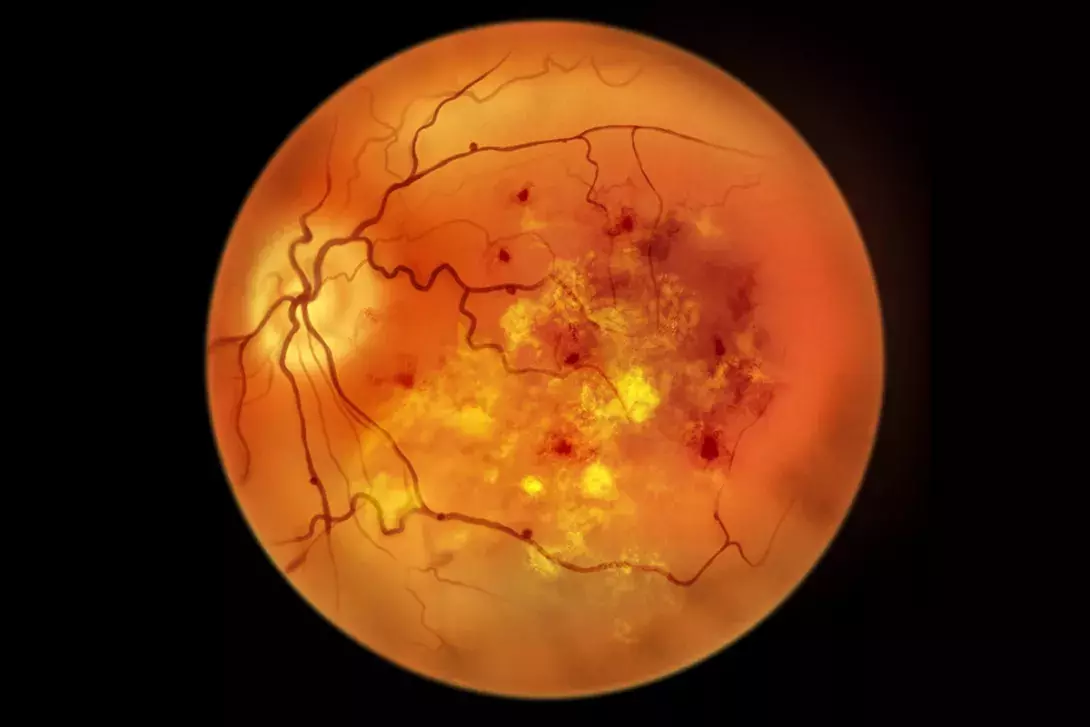
Diabetic retinopathy is a major public health issue in Australia. Patients with diabetes are 25 times more likely to become blind and diabetic retinopathy is the leading cause of vision loss in working-age adults (aged 20 to 74 years). Early detection and treatment of diabetic retinopathy is crucial to prevent vision loss. GPs have an important role in encouraging guideline-recommended screening and should use the specific Medicare Benefits Schedule items for retinal photography that are available for people with diabetes.
- The prevalence of diabetic retinopathy is estimated to rise as rates of diabetes and obesity continue on an upward trajectory.
- Medicare Benefits Schedule items are available specifically to support investigations for retinal photography for people with diabetes; however, these item numbers are underused because of barriers relating to image acquisition and interpretation in general practice.
- New imaging technologies, such as ultrawide field colour fundus and optical coherence tomography imaging as well as optical coherence tomography angiography, can help improve diagnosis of diabetic retinopathy.
- Long-term glycaemic control is the most important factor in reducing the risk of retinopathy and can be achieved through management of lifestyle factors and pharmacotherapy.
About 2.5 to 3 million people in Australia are predicted to have diabetes by 2025, if current obesity and diabetes incidence trends continue upward, with the figure closer to 3.5 million by 2033.1 This will significantly impact the healthcare and economic burden of diabetic retinopathy. According to Australia’s Eye Health Survey 2016, 39.4% of adults with self-reported diabetes aged 40 years of age and older had diabetic retinopathy.2 Regular screening, early identification and appropriate intervention for diabetic retinopathy may prevent up to 90% of diabetes-related vision loss; however, due to poor screening, the prevalence of diabetic retinopathy is often underestimated.3,4 This article summarises aspects of screening, diagnosis and management of diabetic retinopathy that are relevant to general practice.
Screening for diabetic retinopathy
National Health and Medical Research Council guidelines recommend that all people with diabetes undergo an eye examination every two years, starting at diagnosis. Higher-risk patients, such as those with poor glycaemic control and longer duration of diabetes, should undergo annual eye examinations, and patients with diabetic retinopathy should undergo more frequent eye examinations.5 However, only 78% of non-Indigenous Australians and 53% of Indigenous Australians adhere to these guidelines.6
In 2016, two new Medicare Benefits Schedule items (MBS item 12325 for the Aboriginal and Torres Strait Islander population and MBS item 12326 for a general population) were introduced to support retinal photography for people with diabetes and image reporting by GPs. Although modelling predicted over 400,000 patients with diabetes would be screened after the introduction of these item codes, real-world data showed that only 8000 people were screened by GPs between 2016 and 2020 using these MBS items.7
Currently, most diabetic retinopathy screening is performed by optometrists and ophthalmologists, despite the fact that GPs are at the frontline in identifying other complications of this chronic disease. Several barriers to delivering diabetic retinopathy screening in Australian general practices have been identified. A low awareness of the MBS codes for diabetic retinopathy screening among GPs can be a potential barrier, with a recent study finding that only three out of every 15 GPs were aware of the codes.4
Although the MBS codes provide financial incentives, the MBS items require the GP to both perform and interpret the retinal images in patients with diabetes but no prior diagnosis of diabetic retinopathy. Both image acquisition and interpretation are perceived as a significant challenge to conducting diabetic retinopathy screening. In countries such as the UK, all adult patients with diabetes attend regular diabetic retinopathy screening, and these images are traditionally graded by humans, which can be resource intensive. Screening for diabetic retinopathy via telehealth (known as teleretinal screening) has been shown to be highly accurate for detecting diabetic retinopathy, and has been successfully implemented in other health programs such as in Singapore.8 Artificial intelligence approaches to grade diabetic retinopathy automatically are also promising and are particularly appealing in areas with limited medical services, such as remote and rural regions, once appropriately validated.9
Cost and access to conventional retinal cameras are also barriers to retinal screening. A pan ophthalmoscope provides a 20 to 25 times wider view than the traditional direct ophthalmoscope in nondilated eyes, allowing examination of the peripheral retina in diabetic retinopathy. It can be more easily used by GPs in clinical practice and has provision for a smartphone adaptor that connects easily and allows storage and transfer of images (Figure 1a). Smartphones can be used with a condenser lens (20-diopter) (Figure 1b) and is a useful way of obtaining retinal photographs but requires more training than pan-ophthalmoscope methods. Non-mydriatic fundus cameras allow photos to be taken without dilating the pupil; however, these can be costly, limiting uptake.
In Australia, KeepSight is a national diabetes eye screening program that aims to help people with diabetes to get their eyes screened regularly. The program is led by Diabetes Australia, and people registered with the National Diabetes Services Scheme receive eye health information and alerts encouraging them to have a diabetes eye check. Healthcare professionals and individuals can register on the website to receive regular reminders (https://www.keepsight.org.au).
New imaging technologies
Fundus fluorescein angiography (FFA) after injection of fluorescein dye has been used to perform detailed visualisation of retinal vasculature to accurately determine the clinical stage of diabetic retinopathy. This invasive test is limited by the risks of severe allergic reactions to the fluorescein dye and yellowish discolouration of the skin for a couple of days after the procedure. Recent advances in ultrawide field (UWF) imaging and its application to FFA photos allows a far greater area of retinal visualisation; it allows the identification of diabetic retinopathy outside of the traditionally used seven-field Early Treatment Diabetic Retinopathy Study (ETDRS) grid.10 Detection of diabetic disease in the peripheral retina is crucial for diagnosis and monitoring treatment response. Examples of UWF imaging are shown in Figure 2.
Optical coherence tomography angiography (OCT-A) offers a noninvasive method of visualising retinal vasculature without intravenous injection of fluorescein dye. It generates three-dimensional (3-D) images based on the motion contrast of circulating blood cells. As well as being noninvasive, it allows visualisation of the fine capillaries of chorioretinal vasculature at different anatomical levels, as well as neovascularisation.
Optical coherence tomography (OCT) uses infrared light to analyse the retinal structure by means of high-resolution tomographic cross-sections, and is a reliable diagnostic and screening tool for diabetic macular oedema. OCT is a noninvasive and quick scan that is easily accessible for patients in Australia. In the past decade, in which anti-vascular endothelial growth factor (VEGF) injections have revolutionised the treatment of diabetic macular oedema, OCT is paramount for the early detection of diabetic macular oedema and assessment of treatment effect (Figure 3).
Diabetic macular oedema
Diabetic macular oedema (DMO) is the most common cause of central vision loss. Intravitreal injections of anti-VEGF agents are the standard of care in the treatment of centre-involving DMO. VEGF is a member of the platelet-derived growth factor (PDGF) family and plays a crucial role as an initial rate-limiting step in physiological angiogenesis.11 The binding of VEGF to its receptors leads to endothelial cell proliferation, and hence new blood vessel growth. VEGF levels have been found to be elevated in the vitreous of patients with DMO.
Several anti-VEGF agents have been investigated and are available for clinical use. These agents include bevacizumab, ranibizumab, aflibercept and, most recently, faricimab that also targets the angiopoietin-2 inflammatory pathway. Ranibizumab, aflibercept and faricimab are listed on the PBS for documented defined DMO with vision loss. Injections are initially given monthly in the loading phase and then the nterval between injections is extended depending on clinical response. Figure 4 shows a patient undergoing the procedure of intravitreal injection. A major disadvantage of intravitreal injections is the frequent and regular clinical visits and injection regimens, which may be a burden for patients who also likely have other comorbidities for which they need to manage appointments, resulting in pressures of taking time off work.
Intravitreal steroids decrease retinal thickness by targeting the inflammatory component of DMO. A slow-release formulation of dexamethasone is listed on the PBS for DMO. However, steroids are second-line agents as they have potential ocular side-effects, including development of cataracts and glaucoma. The use of intravitreal steroids potentially reduces the need for such frequent injections but does require close monitoring for raised intraocular pressure.
The Early Treatment in Diabetic Retinopathy Study from the 1980s showed that a macular laser can reduce the risk of moderate visual loss.12 There is a long-term risk of laser scars expanding and causing atrophy with central visual loss and, therefore, in modern practice, macular laser is reserved for DMO away from the foveal centre. However, newer subthreshold lasers may have a role in treating mild DMO that is relatively close to the foveal centre.
Proliferative diabetic retinopathy
Proliferative diabetic retinopathy (PDR) can cause severe vision loss and blindness. Peripheral retinal ischaemia causing the development of retinal neovascularisation in PDR is treated by pan-retinal photocoagulation (PRP) (Figure 5). About 1000 to 2000 micro-burns are typically made on the retina, which destroys the largely unused, ischaemic extramacular retina and reduces VEGF production, eventually reducing regression of neovascularisation.13
In the clinic, the PRP laser is delivered via a slit-lamp. A green or yellow laser is typically used to produce small, evenly spaced spots to ischaemic retina. The development of new pattern-scanning lasers allows the delivery of multiple laser spots in rapid succession, reducing treatment time with improved patient tolerance. PRP reduces the risk of neovascular glaucoma, vitreous haemorrhage and tractional retinal detachment, which could otherwise result in severe vision loss. However, PRP can reduce night vision and the peripheral field of vision.
Anti-VEGF therapy also has a minor role in the treatment of PDR. Ranibizumab is approved by the TGA for PDR treatment and it is often used in anterior segment neovascularisation and cases of coexisting DMO. Often, patients present with advanced blinding PDR with nonresolving vitreous haemorrhage (preventing laser application) or scar tissue that causes the retina to detach. These patients may proceed to vitrectomy surgery.
Vitreoretinal surgery for advanced diabetic retinopathy
Vitrectomy is a microsurgical procedure which involves the removal of the vitreous gel from the eye. Vitrectomy is indicated in patients with advanced diabetic retinopathy, such as nonresolving vitreous haemorrhage, dense subhyaloid haemorrhage over the macula, tractional retinal detachment threatening the fovea, or a vitreomacular membrane resulting in nonresolving macular oedema.
Technological advances in vitreoretinal surgery
Recent advances in vitrectomy surgery have significantly improved approaches to diabetic vitrectomy. For example, small-gauge instrumentation (25- and 27-gauge) and higher cutting speeds reduce traction on the retina and allow more efficient removal of the vitreous gel; the ports are closer to the tip of the cutter, allowing precise dissection of preretinal membranes. Figure 6 shows a patient who underwent vitrectomy after presenting with a large diabetic vitreous haemorrhage.
Eye surgeons are now able to use modern 3-D heads-up microscopes for diabetic vitrectomy. The surgical microscope eyepieces are replaced with high-resolution stereoscopic cameras transmitting an image to a screen for 3-D viewing. According to its manufacturers, this new visualisation system provides five times the extended depth of field, 48% increased magnification and 42% finer stereopsis compared with traditional microscopes.14 This is particularly useful in diabetic vitrectomy, when visualising and dissection of fine membranes is paramount. In vitrectomy surgery, surgeons may use intraoperative OCT to examine cross-sectional anatomical information in real time and high resolution, allowing accurate identification of membranes on the surface of the retina.
Systemic management
Patients should be counselled on lifestyle measures, especially dietary advice, increasing physical activity and smoking cessation, as these can improve glycaemic control and reduce the risk of retinopathy. Improving key modifiable systemic risk factors, including maintaining good glycaemic control and blood pressure, can prevent or delay diabetic retinopathy.
In some patients, a rapid improvement in blood glucose control can actually lead to an acceleration of diabetic retinopathy in the short term. For example, a potential association between the use of semaglutide and early diabetic retinopathy progression has been suggested.15 Closer ophthalmology review should be considered in patients with sudden improvement in blood glucose level.
Fenofibrate may also have a role in the management of diabetic retinopathy in type 2 diabetes and has been shown to have a beneficial effect in preventing DMO and slowing diabetic retinopathy progression in two clinical trials.16 Although it is primarily a lipid-lowering drug, the mechanism of action of fenofibrate in diabetic retinopathy appears to be independent of serum lipid concentration. Caution is required when prescribing it to patients with renal or hepatic impairment and the risk of myositis must be considered. Interpretation of the evidence for fenofibrate and its use in the medical community globally has been variable. According to the TGA, in Australia, ‘fenofibrate is indicated for the reduction in the progression of diabetic retinopathy in patients with type 2 diabetes and existing diabetic retinopathy’.17
If diabetic retinopathy results in significant vision loss, patients should be linked with appropriate support systems. For example, Vision Australia has referral services to appropriate specialists who can assess and provide appropriate low-vision aids and training. Financial support may also be available for mobility and transport, education and support at work for patients with low vision.
Conclusion
Early screening and diagnosis for diabetic retinopathy is crucial to prevent vision loss in the 1.5 million people in Australia living with diabetes. GPs can play an important role in encouraging people with diabetes to be screened regularly for diabetic retinopathy, for example, by registering with KeepSight. Good long-term glycaemic control is the most important factor in reducing the risk of sight-threatening retinopathy. Therefore, early and close communication between the patient, GP, ophthalmologists and optometrists can also improve patient outcomes, as vision-threatening complications tend to have better outcomes if treated early and proactively. An awareness of available support for patients with diabetes and who have vision loss is important. ET
COMPETING INTERESTS: Dr Mehta has received speaker fees from AbbVie, Apellis, Bayer and Roche; travel support from Bayer; and been on Advisory Boards for AbbVie, Apellis and Roche. Associate Professor Chang has received consultancy fees from Alcon, Apellis, Bayer, Roche and Novartis; speaker fees from Alcon, Apellis, Bayer and Roche; Travel support from Bayer and Roche; is the Secretary General Asia Pacific Vitreoretinal Society; and Chair of the Board, Sydney Eye Hospital Foundation. Dr Ham: None.
